Pfizer 2012 Annual Report Download - page 87
Download and view the complete annual report
Please find page 87 of the 2012 Pfizer annual report below. You can navigate through the pages in the report by either clicking on the pages listed below, or by using the keyword search tool below to find specific information within the annual report.
Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
86
2012 Financial Report
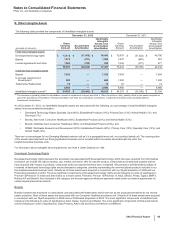
In-Process Research and Development
IPR&D assets represent research and development assets that have not yet received regulatory approval in a major market. The more
significant components of IPR&D are a treatment for skin fibrosis and programs for the treatment of staph aureus infections and epilepsy, as
well as a vaccine for the prevention of meningitidis serogroup B in adolescents and young adults.
IPR&D assets are required to be classified as indefinite-lived assets until the successful completion or the abandonment of the associated
research and development effort. Accordingly, during the development period after the date of acquisition, these assets will not be amortized
until approval is obtained in a major market, typically either the U.S. or the EU, or in a series of other countries, subject to certain specified
conditions and management judgment. At that time, we will determine the useful life of the asset, reclassify the asset out of in-process
research and development and begin amortization. If the associated research and development effort is abandoned, the related IPR&D assets
will likely be written-off, and we will record an impairment charge.
Among the IPR&D assets reclassified to Developed Technology rights as a result of being approved in a major market were the following: in
2012, two IPR&D assets with a combined book value of approximately $160 million and, in late 2011, Prevenar 13 for adults age 50 years and
older and Vyndaqel (tafamidis meglumine), with a combined book value of approximately $2.3 billion.
For information about impairments of IPR&D assets, see Note 4. Other Deductions––Net.
For IPR&D assets, the risk of failure is significant and there can be no certainty that these assets ultimately will yield a successful product. The
nature of the biopharmaceutical business is high-risk and, as such, we expect that many of these IPR&D assets will become impaired and be
written off at some time in the future.
Amortization
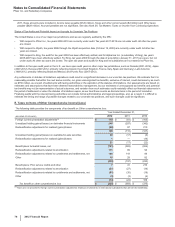
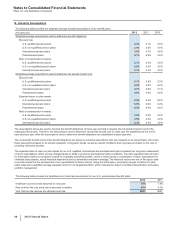
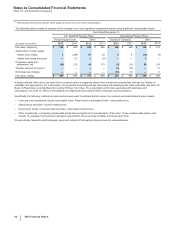
The weighted-average life of both our total finite-lived intangible assets and the largest component, Developed technology rights, is
approximately 11 years. Total amortization expense for finite-lived intangible assets was $5.4 billion in 2012, $5.8 billion in 2011 and $5.5
billion in 2010.
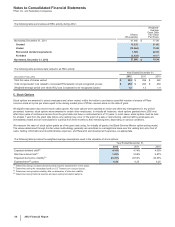
The following table provides the annual amortization expense expected for the years 2013 through 2017:
(MILLIONS OF DOLLARS) 2013 2014 2015 2016 2017
Amortization expense $4,804 $4,145 $3,735 $3,488 $3,373
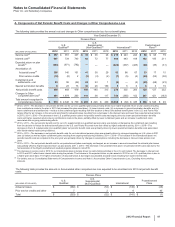
Note 11. Pension and Postretirement Benefit Plans and Defined Contribution Plans
The majority of our employees worldwide are covered by defined benefit pension plans, defined contribution plans or both. In the U.S., we
have both qualified and supplemental (non-qualified) defined benefit plans. A qualified plan meets the requirements of certain sections of the
Internal Revenue Code, and, generally, contributions to qualified plans are tax deductible. A qualified plan typically provides benefits to a broad
group of employees with restrictions on discriminating in favor of highly compensated employees with regard to coverage, benefits and
contributions. A supplemental (non-qualified) plan provides additional benefits to certain employees. In addition, we provide medical and life
insurance benefits to certain retirees and their eligible dependents through our postretirement plans.
Beginning on January 1, 2011, for employees hired in the U.S. and Puerto Rico after December 31, 2010, we no longer offer a defined benefit
plan and, instead, offer an enhanced benefit under our defined eligible contribution plan. In addition to the standard matching contribution by
the Company, the enhanced benefit provides an automatic Company contribution for such eligible employees based on age and years of
service.
On May 8, 2012, we announced to employees that as of January 1, 2018, Pfizer will transition its U.S. and Puerto Rico employees from its
defined benefit plans to an enhanced defined contribution savings plan. As a result of this decision to freeze the U.S. and Puerto Rico defined
benefit plans, a curtailment was triggered and we performed a re-measurement of the pension obligations and plan assets in the second
quarter of 2012, which had an immaterial impact to the funded status of the plans. For the year ended December 31, 2012, we recorded,
among other impacts, a curtailment gain of approximately $59 million in the consolidated statement of income.